
Features
What are the key elements to promote and ensure fish welfare in a RAS environment?
June 21, 2021 By Dr Åsa Maria Espmark
 Nofima researcher Khurram Shahzad is securing velocity and distribution with computational fluid dynamic. Photo: Terje Aamodt
Nofima researcher Khurram Shahzad is securing velocity and distribution with computational fluid dynamic. Photo: Terje Aamodt To farm fish in recirculating aquaculture systems requires skilled and dedicated employees. RAS systems are filled with technologies that need to be managed well to fulfill fish health and welfare. When done correctly, RAS has many advantages, also for fish welfare. Maybe the biggest advantage is the possibility to have full control of the system and water quality, and hence fish welfare and performance. This advantage can also turn out to be the contrary if water quality and housing conditions are not properly taken care of. If we are considering the use of RAS for post-smolt or even land-based to market size, we also have the advantage of keeping the fish longer away from open sea, thus longer away from sea lice and escapee. Additionally, this gives the possibility to produce salmon in areas not otherwise suited for aquaculture, leading to locally produced food and jobs.
At Nofima, we are hosting CtrlAQUA, a centre for research-based innovation aiming towards biological and technological innovations to further develop RAS and semi-closed containment solutions for production of salmon post-smolt. Welfare is central in CtrlAQUA and there is much research to promote. In this short article I will give a few examples.
Avoid deadly gas
To be able to utilise the advantages with RAS and ensure good welfare, there are some challenges that need to be solved. The formation of hydrogen sulfide has for long been a huge threat in RAS, with the risk of massive and acute mortalities. Significant research to establish analysing methods, identify limit values and early warning markers, and determine long-term effects of sublethal concentrations are on-going. In addition, good practices of how to avoid formation of hydrogen sulfide are also being produced.
Find the right water quality for RAS
As already mentioned, good water quality is one key factor for good welfare. It has been shown that there are needs for RAS-specific limit values for essential water quality variables, as many recommended limit values are still based on research performed in flow-through (FT) systems, and since some of them may differ between FT and RAS. Mota et al., (2019) demonstrated this for carbon dioxide, and showed a growth penalty with CO2 concentrations from 5 to 40 mg/l (Figure 1), without any severe welfare effects. However, the effect on growth already at lower concentrations than the recommended 15 mg/l, suggests that in RAS the CO2 levels should be kept even lower. The authors suggest 12 mg/l in RAS (Mota et al., 2019).
Be more careful with ozone in saline water
To maintain good water quality in RAS, water treatment is needed to remove unwanted particles and gasses. In CtrlAQUA ongoing research deal with biofilters, CO2 degassing, UV and ozone treatment, in addition to disinfection strategies. Many farmers prefer to treat the RAS water with ozone to reduce the coloration and increase the visibility in the water. However, due to the formation of bromines in ozone-treated saline water, that may be toxic for the fish. The risk of using ozone in saline water is higher than to use it in freshwater. In freshwater, ozone significantly improved water clarity, diminished bacteria counts, and reduced dissolved metals. It can also lead to increased salmon growth (Davidson et al., 2021). On the contrary, salmon is sensitive towards ozone in saline water, and Stiller et al., (2020) demonstrated that ozone concentrations of 500 mV (44 µg as l-1 as Cl2), that it just below what is recommended for turbot (Scophthalmus maximus) and sea bass (Dicentrarchus labrax), turned out to be acutely lethal for salmon and was especially harmful for the gills. For salmon in brackish water, 350 mV (10 µg as l-1 as Cl2) is considered safe (Stiller et al., 2020).
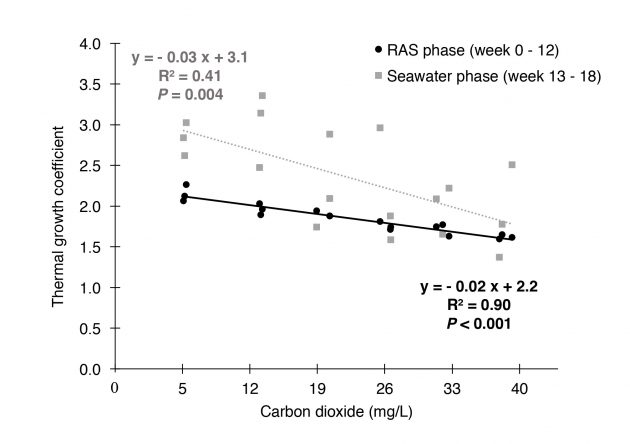
Figure 1. Growth penalty caused by CO2 during the RAS phase was carried over to the flow through on-growing phase (Mota et al., 2019).
Give the fish possibilities for exercise
Exercise in fish has proven to be beneficial for welfare, and a lot of research has concluded the same (e.g. Totland et al., 2011). One way to facilitate training is with water velocity (Castro et al., 2011), and there has been a need to define water velocities that provide the optimal swimming speed for exercise in salmon in RAS. Timmerhaus et al., (2020) exposed salmon post-smolt to velocities of 0.5, 1.0, 1.8 and 2.5 BL/sec. It was seen that the weight increased linearly with velocity. It was also a linear increase in muscle cell density, indicating that the weight increase was indeed an increase in muscle mass, caused by exercise. However, the welfare of the fish at the highest velocity (2.5 BL/sec) was not good, as shown by the increased number of severe skin damages. Also, schooling was observed after a few days with the highest velocities. The reason for this behavioural change is not proven, but the assumption may be because the fish tried to form some hiding places from velocities. It may also be because possible uneven distribution of velocities in the tanks resulted in places with lower velocities that were more preferred places for the fish than the higher velocity areas. From this study it was concluded that recommended swimming speed in RAS for post-smolt is 1.0 to 1.5 BL/sec (Timmerhaus et al., 2020).
The image on page 14 shows a snapshot of welfare research performed in CtrlAQUA for salmon post-smolt. Most of them are also relevant for market size production, but for land-based to slaughter also other welfare issues may arise. You are welcome to visit the website (www.ctrlaqua.no) for more information about the research done in the centre.
(For a list of literature referred to in this article, please email the editor at mdeguzman@annexbusinessmedia.com)
Dr Åsa Maria Espmark is a senior scientist in Nofima, Norway. For the last 20 years her scientific interests have been related to welfare, welfare indicators and stress, both in Atlantic salmon and the cleaner fish species lump fish and Ballan wrasse. Espmark has been coordinating several national and international projects, like the EU projects OrAqua and FutureEUAqua. Since October 2017, she has been the Center Director for CtrlAQUA SFI, partly funded by Research Council of Norway, aiming at developing technological and biological innovations to make closed containment systems a reliable and economic sustainable technology.
Print this page